Crystal Miner Software PCR Analysis
Get results you can see with user-friendly software for digital PCR analysis
Crystal Miner software
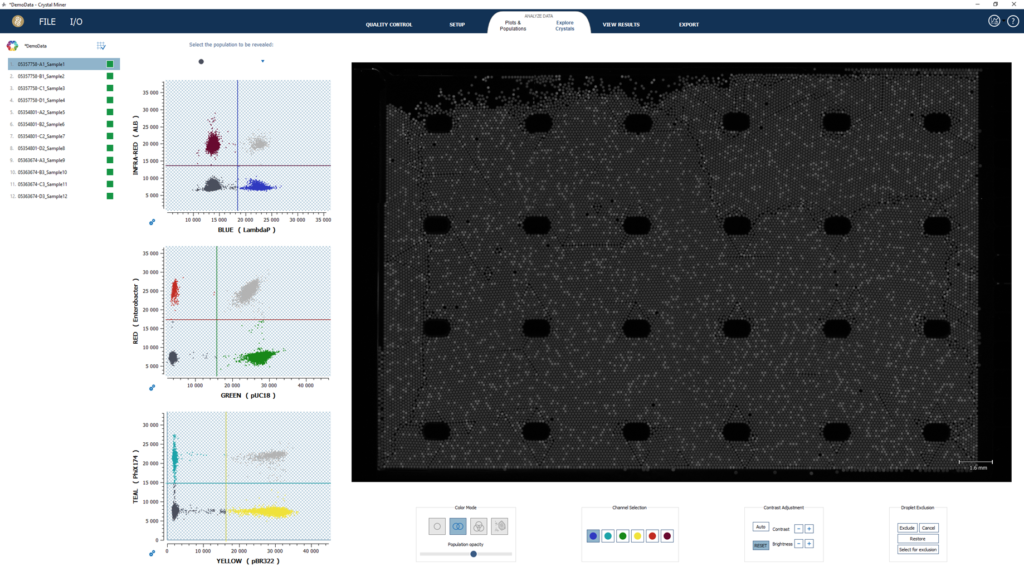
Crystal Miner software automatically identifies positive and negative droplets in all the fluorescence channels of interest and measures the concentrations of targeted nucleic acids.
With its intuitive visuals, Crystal Miner software eases the analysis of complex and highly multiplexed assays.
The unique Explore Crystal feature linking any dot in a dot plot to its corresponding droplet in the Droplet Crystal allows for a comprehensive analysis of the dot plots and provides precious information on assay performances for a better control of the quality of the experiment.
Along with the ability to read data arranged along 1D, 2D, and 3D dot plots, it is possible to get access to raw data and export experimental details and results.
| License & update fees | Free |
| File format | .ncx (naica® crystal experiment) .ncr (naica® crystal result) |
| Data visualization | Droplet Crystal images; 1D, 2D, 3D plots |
| File size | 60 MB / Sapphire Chip chamber (.ncx) 15 MB / Sapphire Chip chamber (.ncr) 30 MB / Ruby Chip chamber (.ncx) 7.5 MB / Ruby Chip chamber (.ncr) |
| Export format | .csv / .xslx / .png / .yaml |
| Threshold | Automatic and manual |
| Compensation matrix | Automatic and manual |
| Pooling | Manual |
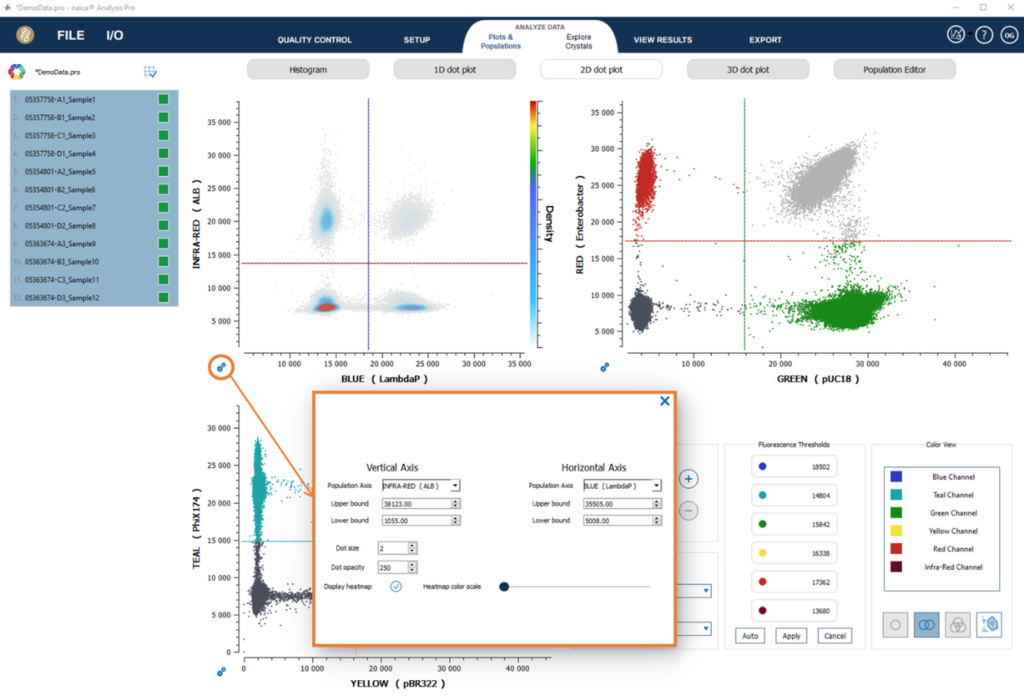
Heatmap visualization
The heatmap visualization is a convenient analysis tool to delimit 2D zones. In heatmap visualization mode, the colormap represents the different droplet density levels in the fluorescence populations. Droplet densities are visualized utilizing a color gradient displayed on the second y axis. Low droplet densities are represented by shades of colours on the lower axis (light grey) while increasing droplet densities are color coded in shades of blue, green, yellow and red representing the highest droplet density.
To activate the heatmap visualization in 2D dot plots, open plots settings and check the “Display heatmap” checkbox.
The color scale on the right of the graph gives the complete color range that is used to represent the different density levels.
Most of the time, the negative population is very dense, which has the effect of hiding smaller density variations between less dense areas. The Heatmap color scale parameter can be modified to highlight populations of lower density levels.
naica® system Pro software (for 21 CFR Part 11 compliance)
An increasing number of laboratories are using electronic records and electronic signatures for exchanging and storing data. Electronic documentation offers many benefits, including increased efficiency and productivity when storing data and easier information sharing and data mining.
The Food and Drug Administration (FDA) of the United States (USA) regulates the food and drug industry of the USA with the Code of Federal Regulations (CFR). The FDA is within the Department of Health and Human Services and manages the Center for Devices and Radiological Health.
If a user’s organization or laboratory intends to use an electronic format instead of paper for records that are required under FDA regulations and requirements, the user´s organization or laboratory must comply with the regulations issued by the FDA: Final Rule 21 CFR Part 11 Electronic Records.
Title 21 of the CFR describes the requirements and regulations for the food and drug industries. For food and drugs to be used within the USA, they must comply with the requirements of this regulatory body. Manufacturers must register with the FDA and obtain approval for a license to distribute their product within the USA. Internationally, FDA regulatory compliance is recognized as a benchmark for the pharmaceutical industry with respect to research, drug development, drug manufacture and sales and marketing of pharmaceutical products.
Part 11 of Title 21 of the Code of Federal Regulations (referred to as 21 CFR Part 11) ‘Electronic Records; Electronic Signatures’ states the rules, definitions, and guidelines under which the FDA, “considers electronic records, electronic signatures, and handwritten signatures executed to electronic records to be trustworthy, reliable, and generally equivalent to paper records and handwritten signatures executed on paper.” The rule defines a standard under which an organization must operate to comply with 21 CFR Part 11.
The naica® system Pro is a closed system, where access is controlled by users who are responsible for the content of the electronic records on that system.
The naica® system Pro software applications, described below, form parts of the ER system by which electronic records are created, modified, stored, and secured against further modification.
The naica® system Pro further provides electronic signature functionality.
The naica® system Pro software includes the following products:
- naica® Data Service
naica® Data Service functions as the naica® system user account manager and controls the compliance of all naica® system operations with respect to 21 CFR Part 11 regulation.
- naica® Reader Pro
naica® Reader Pro software application functions as the operating interface to define the experimental settings for the image acquisition on the scanner instrument (Prism3 / Prism6) for the Crystal Digital PCR™ workflow.
- naica® Analysis Pro
naica® Analysis Pro software is the data analysis application of the naica® system. naica® Analysis Pro is used to analyze data from the images acquired using the naica® Reader Pro, and to calculate the absolute concentrations of targeted nucleic acids.
For Research Use Only. Not for use in diagnostic procedures.
Have a question?
Looking to learn more about this, or any other product in our catalog?